FGFR inhibitors in cholangiocarcinoma: what's now and what's next? - Anna Saborowski, Ulrich Lehmann, Arndt Vogel, 2020
Cureus | Current Targeted Therapy Options in the Treatment of Cholangiocarcinoma: A Literature Review | Article
Open-label, non-randomized, exploratory pre-operative window-of-opportunity trial to investigate the pharmacokinetics and pharma
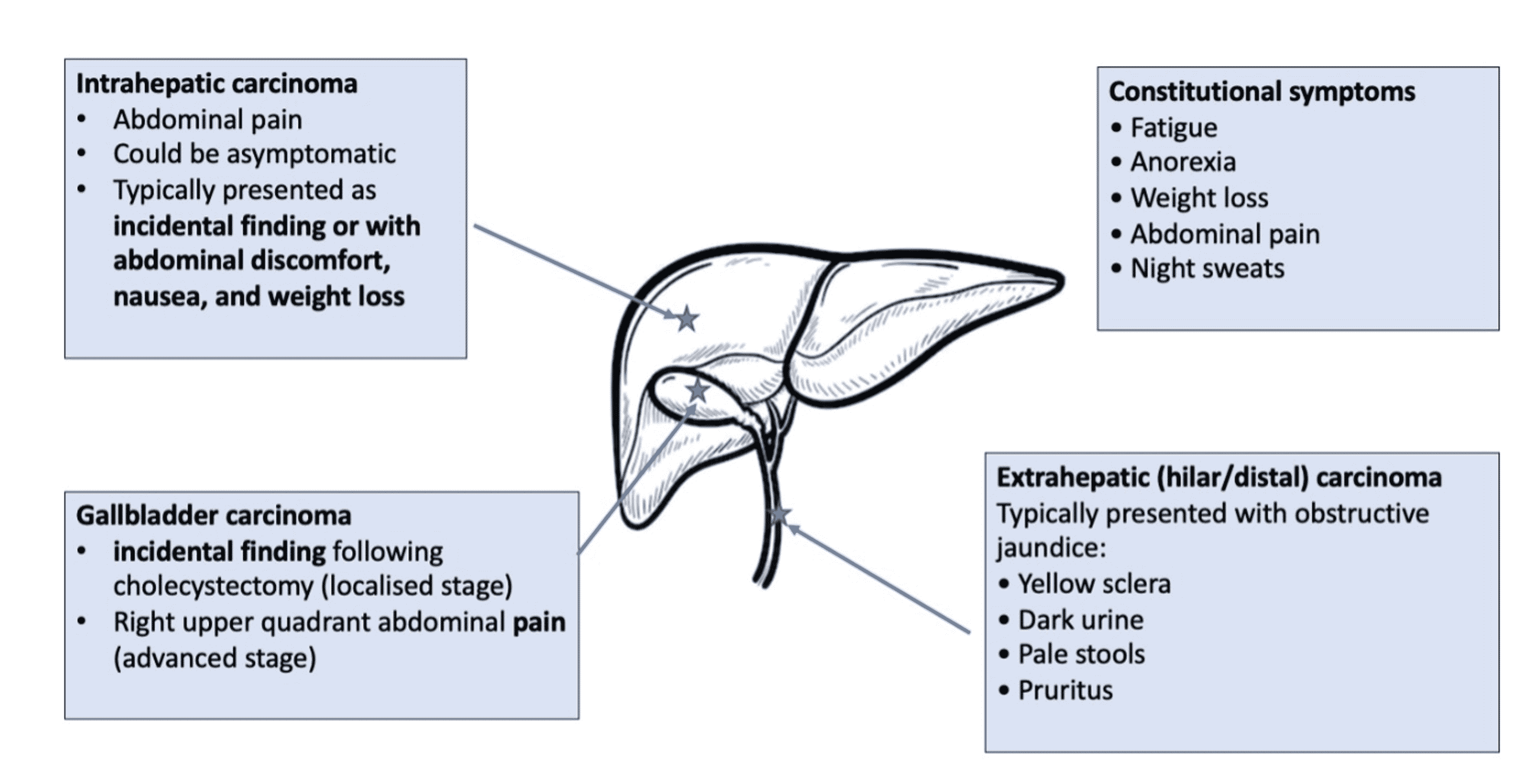
Cureus | Current Targeted Therapy Options in the Treatment of Cholangiocarcinoma: A Literature Review | Article
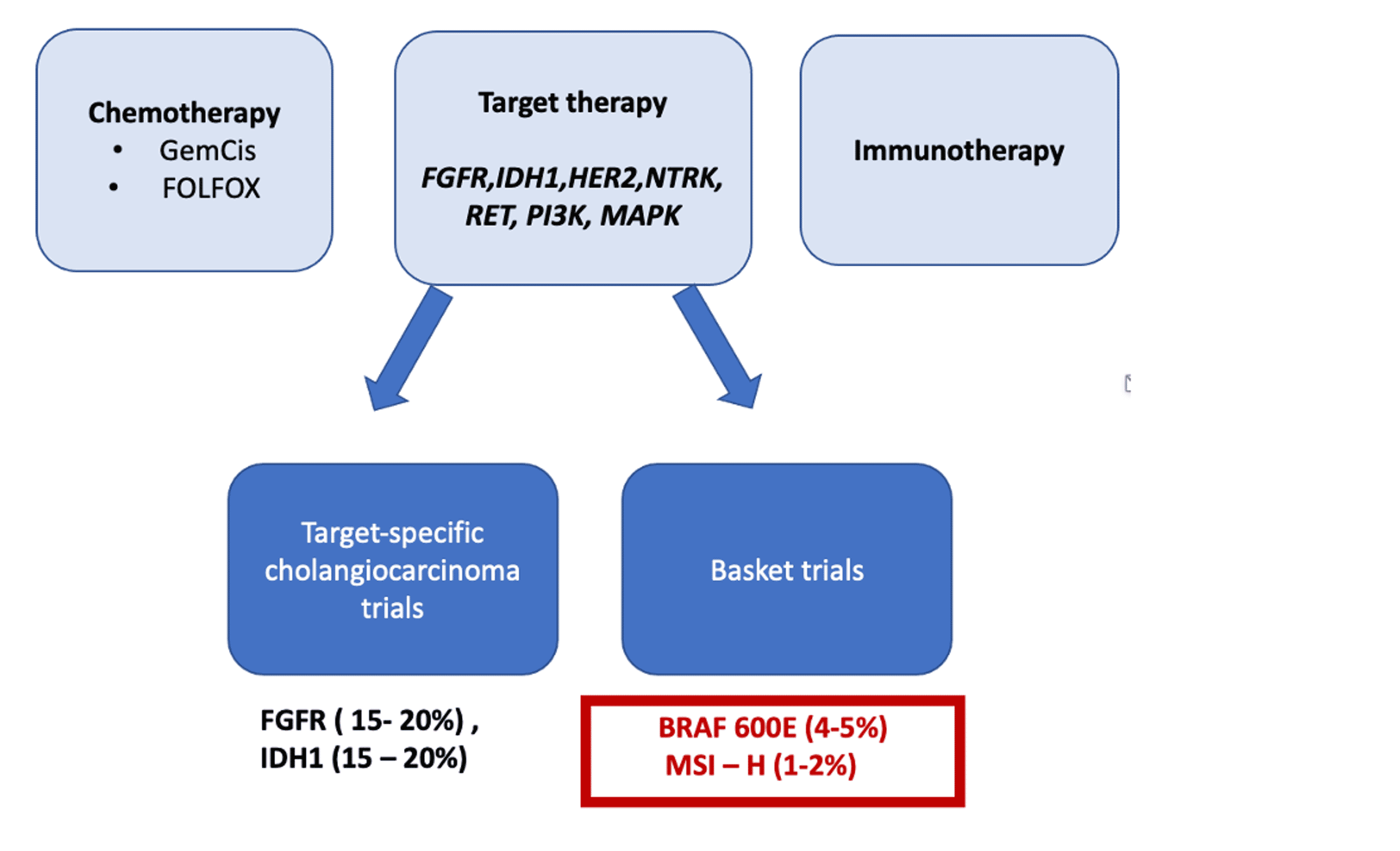
Cureus | Current Targeted Therapy Options in the Treatment of Cholangiocarcinoma: A Literature Review | Article

Conceptual Framework for Therapeutic Development Beyond Anti–PD-1/PD-L1 in Urothelial Cancer | American Society of Clinical Oncology Educational Book

FGFR inhibitors in cholangiocarcinoma: what's now and what's next? - Anna Saborowski, Ulrich Lehmann, Arndt Vogel, 2020
Caris Life Sciences Partners with Debiopharm International SA to Develop Companion Diagnostic for Detection of Rare FGFR Fusions

PDF) 560P Safety and efficacy of Debio 1143, an antagonist of inhibitor of apoptosis proteins (IAPs), in combination with nivolumab in a phase Ib/II trial in patients (pts) failing prior PD-1/PD-L1 treatment
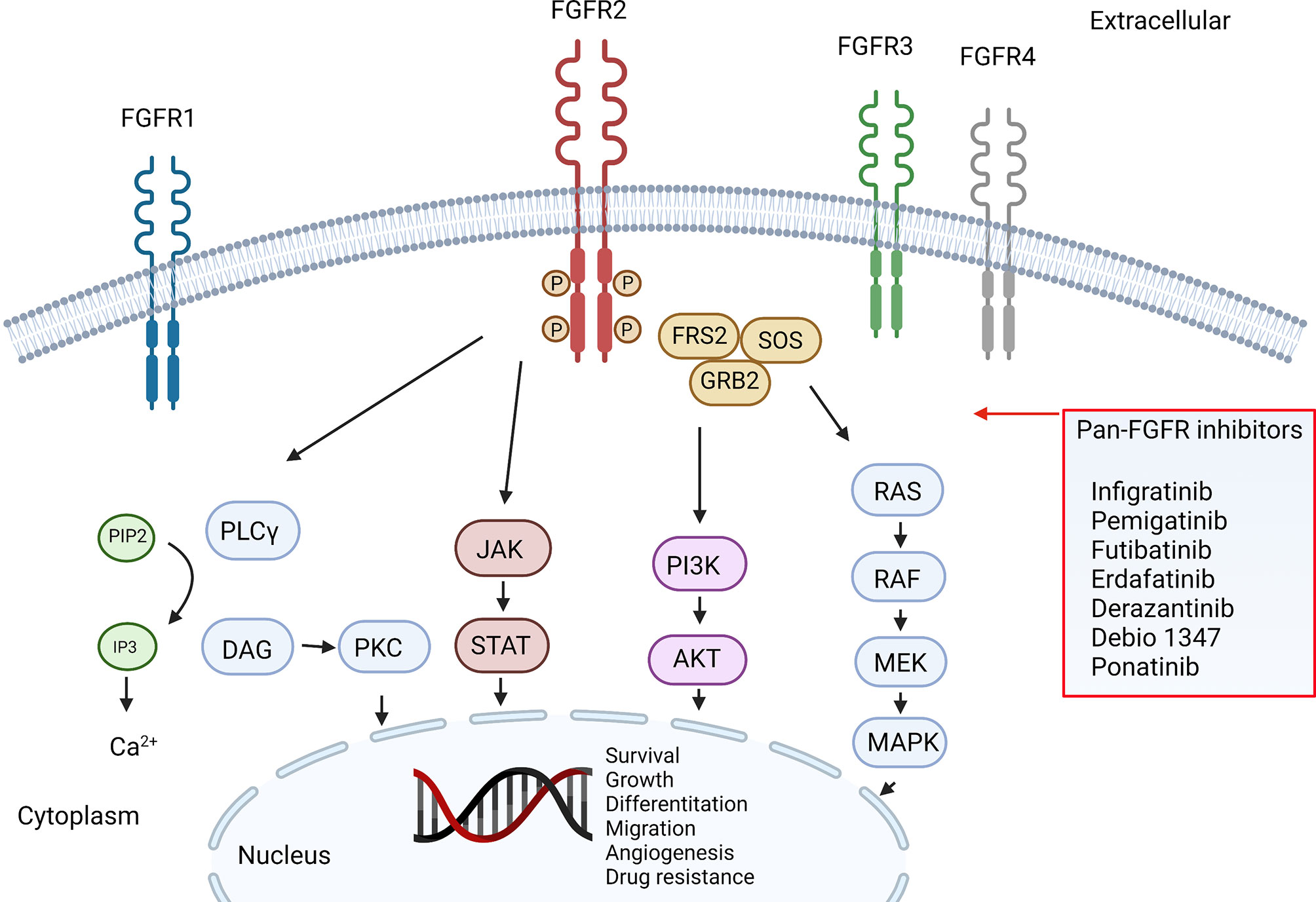
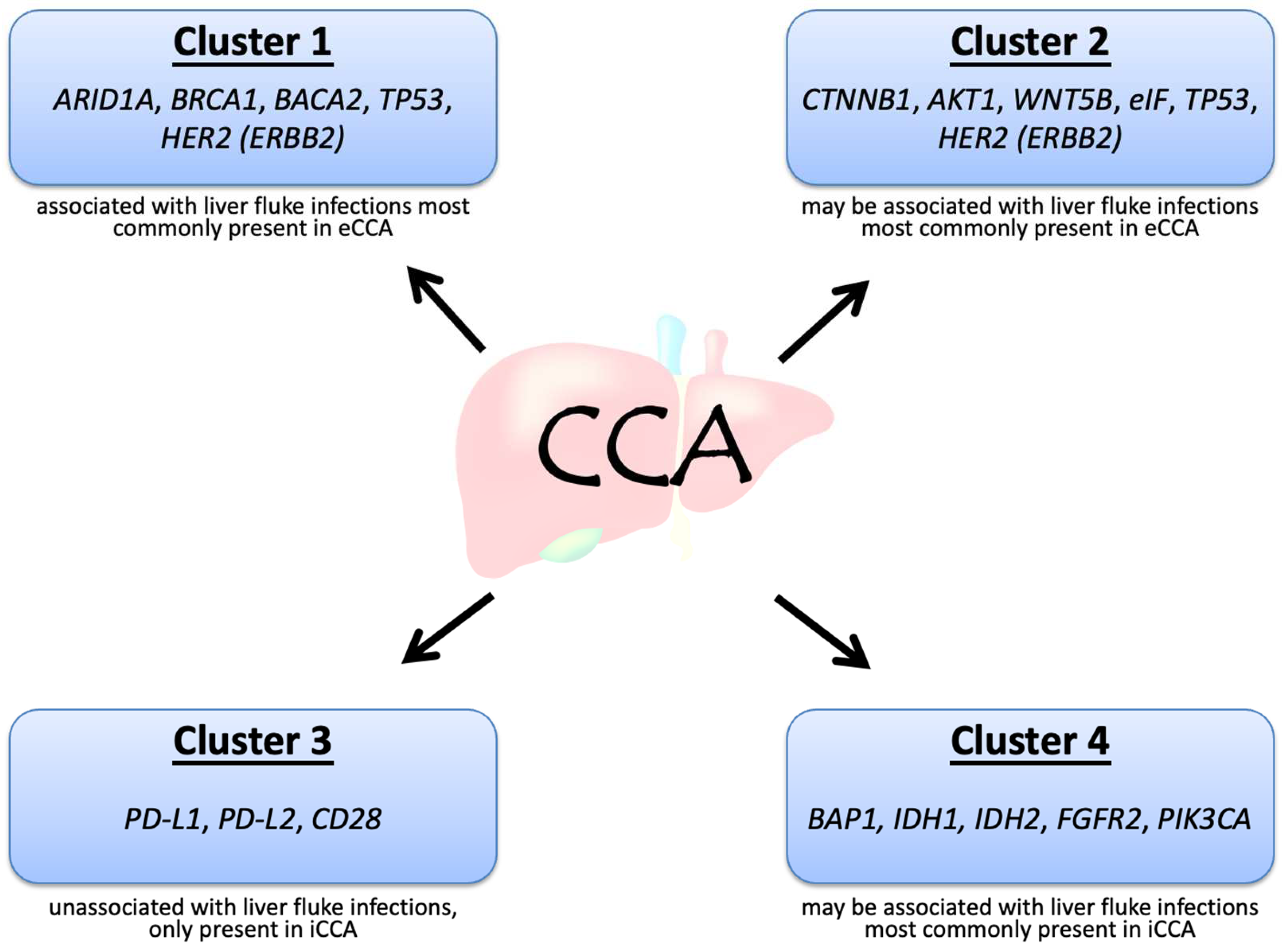
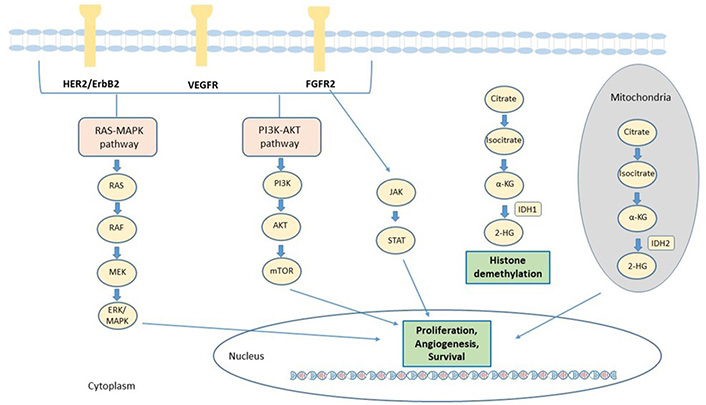
Frontiers | Precision Medicine Targeting FGFR2 Genomic Alterations in Advanced Cholangiocarcinoma: Current State and Future Perspectives

Pertuzumab and trastuzumab for HER2-positive, metastatic biliary tract cancer (MyPathway): a multicentre, open-label, phase 2a, multiple basket study - The Lancet Oncology
Open-label, non-randomized, exploratory pre-operative window-of-opportunity trial to investigate the pharmacokinetics and pharma
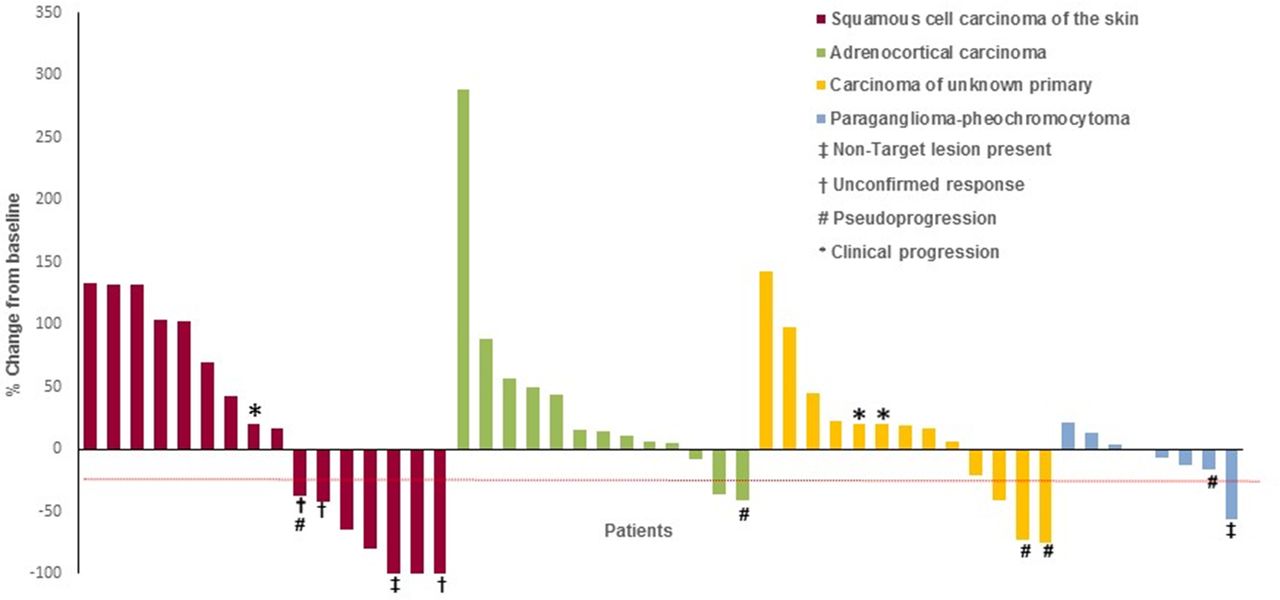
Phase 2 study of pembrolizumab in patients with advanced rare cancers | Journal for ImmunoTherapy of Cancer
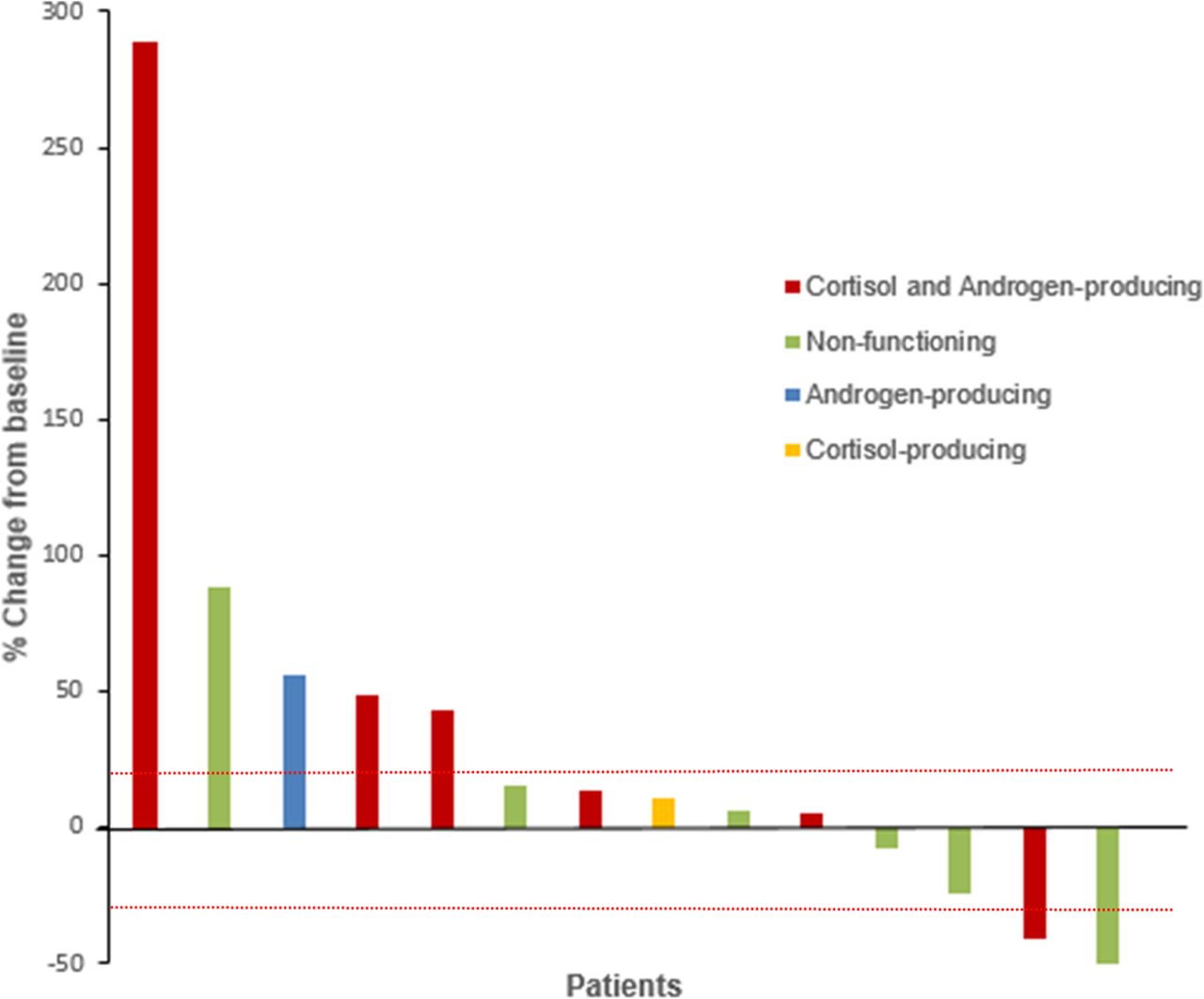
Phase II clinical trial of pembrolizumab efficacy and safety in advanced adrenocortical carcinoma | Journal for ImmunoTherapy of Cancer
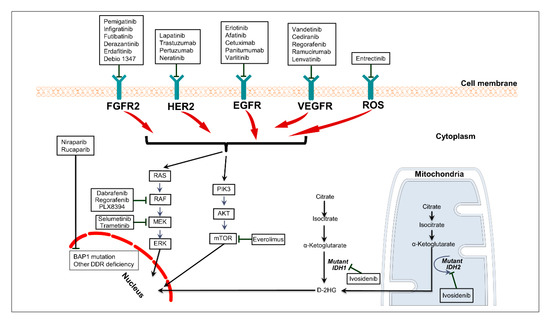
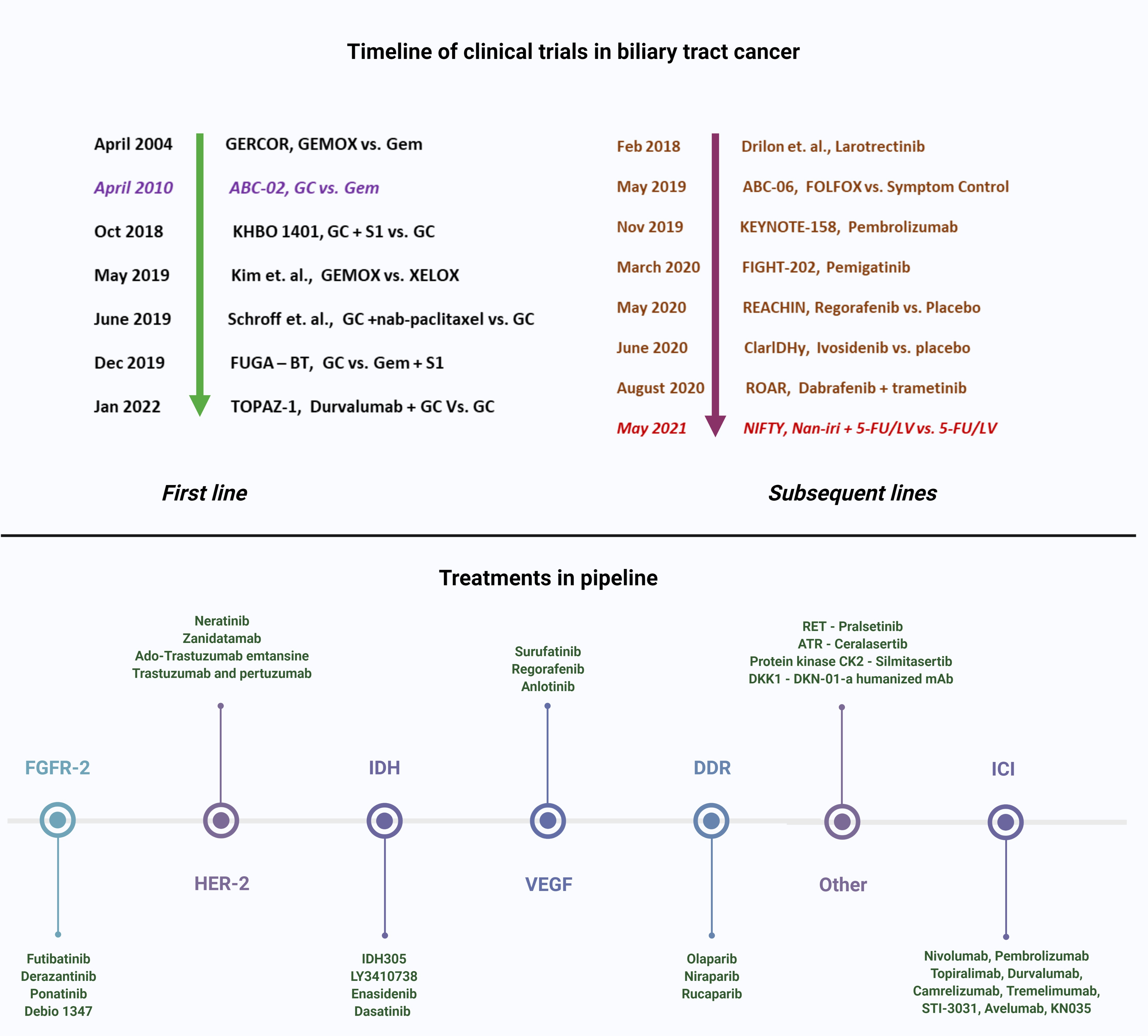
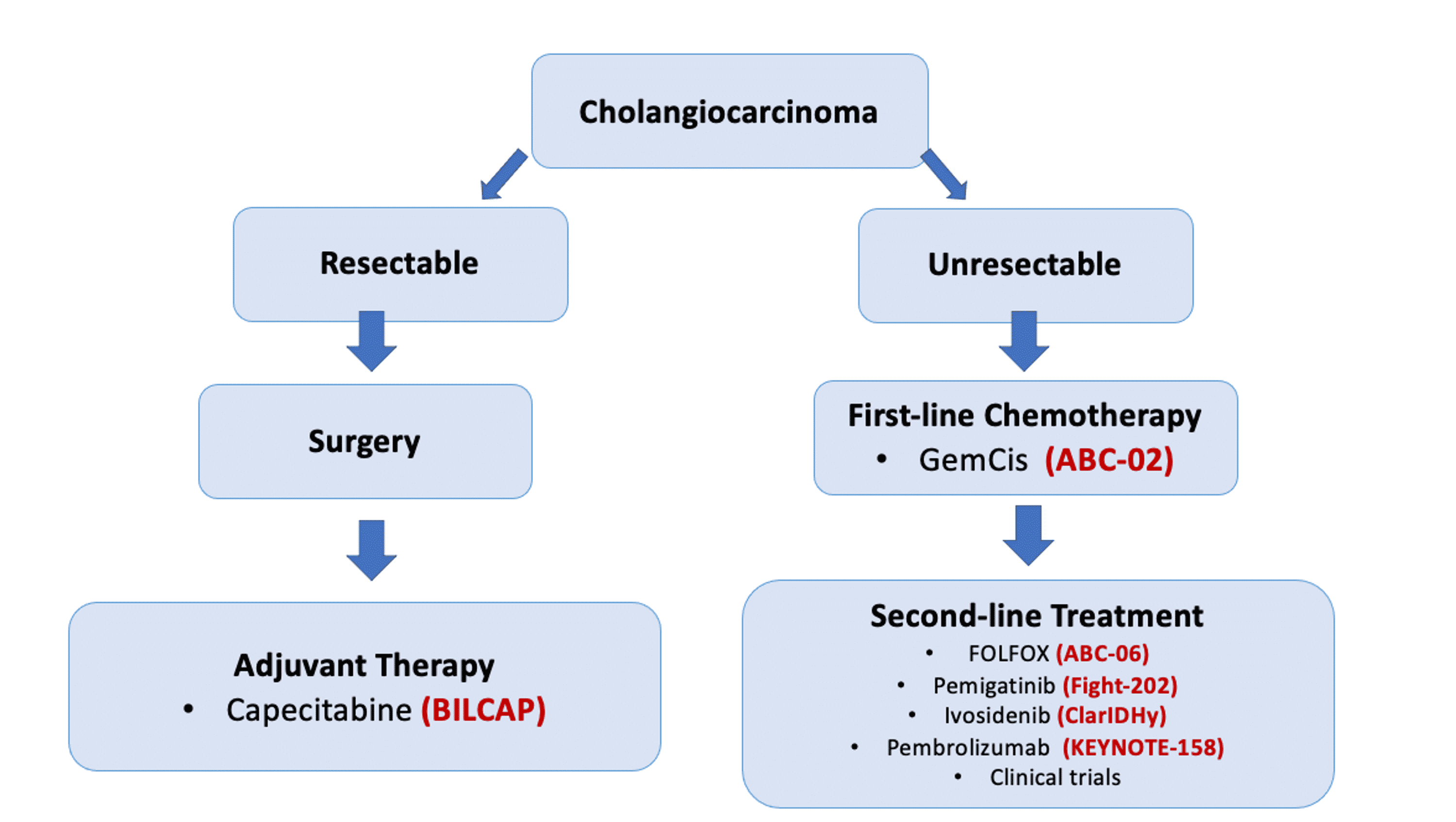
Cancers | Free Full-Text | Targeted Therapies in Advanced Biliary Tract Cancer: An Evolving Paradigm
Open-label, non-randomized, exploratory pre-operative window-of-opportunity trial to investigate the pharmacokinetics and pharma